Jan Demel
Research fellow
Institute of Inorganic Chemistry of the Czech Academy of Sciences
Selected publications
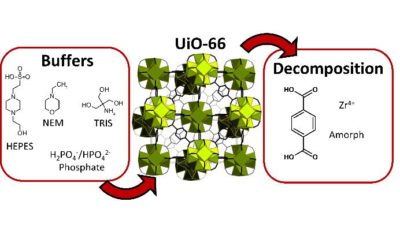
Metal–organic frameworks vs. buffers: case study of UiO-66 stability
Inorg. Chem. Front. 2021
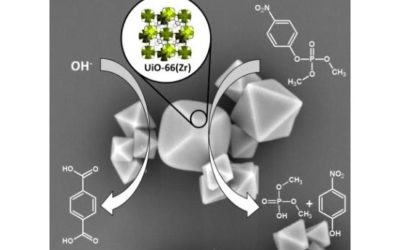
UiO-66 – stability in aqueous environment and its relevance for organophosphate degradations
Inorg. Chem. 2018
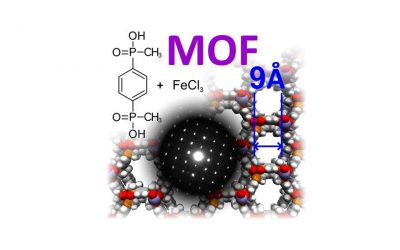
Phosphinic Acid Based Linkers: Building Blocks in Metal–Organic Framework Chemistry
Angew. Chem. 2018, 130, 5110 –5113
Research
Porous coordination polymers – MOFs
Metal-organic frameworks (MOFs) are novel class of crystalline materials composed of metal centres (secondary building units, SBU) connected by organic linkers via coordination bond. The tunability of pore size, crystal topology, and chemical nature of the structures make MOFs candidates for applications like gas storage and separation, heterogeneous catalysis, sensors, drug delivery systems, and many others.
One of the drawbacks of many MOFs is their low stability that comes from the lability of the coordination bonds. For this reason we focus on the preparation of novel MOFs with high hydrothermal stability.
Our approach uses linkers bearing phosphinic acid groups that we combine with hard metals (according to Pearson’s HSAB theory, e.g., Fe3+) for the construction of MOFs which results in high hydrothermal stability of the resulting MOFs. The first examples are MOFs ICR-2 and ICR-4.
Additionally, we study the mechanisms of MOF degradation and the use of novel MOFs for potential applications.
Organic-inorganic hybrid materials for photophysical applications
Composite materials formed by the combination of inorganic and organic materials are attractive for the purpose of combining complementary properties. Of particular interest are materials where this combination is achieved at the molecular level forming ‘organic—inorganic hybrid materials’.
We are interested in incorporation of porphyrins into inorganic matrixes for photophysical applications. Because porphyrins tend to aggregate via π-π interactions which lead to fast quenching of porphyrin excited states and the loss of photophysical properties separation of porphyrin units has to be ensured in order to preserve the photophysical properties. For this reason we study systems with good control over the close environment of porphyrins, mainly layered hydroxides with intercalated porphyrins and MOFs containing porphyrin units.
Such materials then produce singlet oxygen (O2(1Δg)) which can be used for bactericidal coatings or photodynamic therapy.